A new study shows that brain images of cognitively normal adults in their mid-50s can help in predicting the progression of mild cognitive impairment more than a decade later.
Mild cognitive impairment is a sign of early memory loss or other cognitive disabilities and can be a precursor to Alzheimer’s disease, one of the most common forms of dementia.
The results appeared in the journal “Brain Communications“ in July 2021.
Dr. William Kremen, a neuropsychologist who is affiliated with the Center of Excellence for Stress and Mental Health at VA San Diego, led the study. He embarked on the research because little is known about the ability of brain images of cognitively normal adults to predict progression to mild cognitive impairment. Most of the research on brain images in the study of Alzheimer’s disease – referred to as Alzheimer’s disease signatures in the paper – has involved adults over 70 years of age.
“The pathological process in Alzheimer’s disease begins 20 or more years before dementia onset,” says Kremen, who is also a professor of psychiatry at the University of California, San Diego. “Identifying risk factors in cognitively unimpaired middle-aged adults may be a step toward earlier intervention.”
Veterans in study averaged 56 years of age
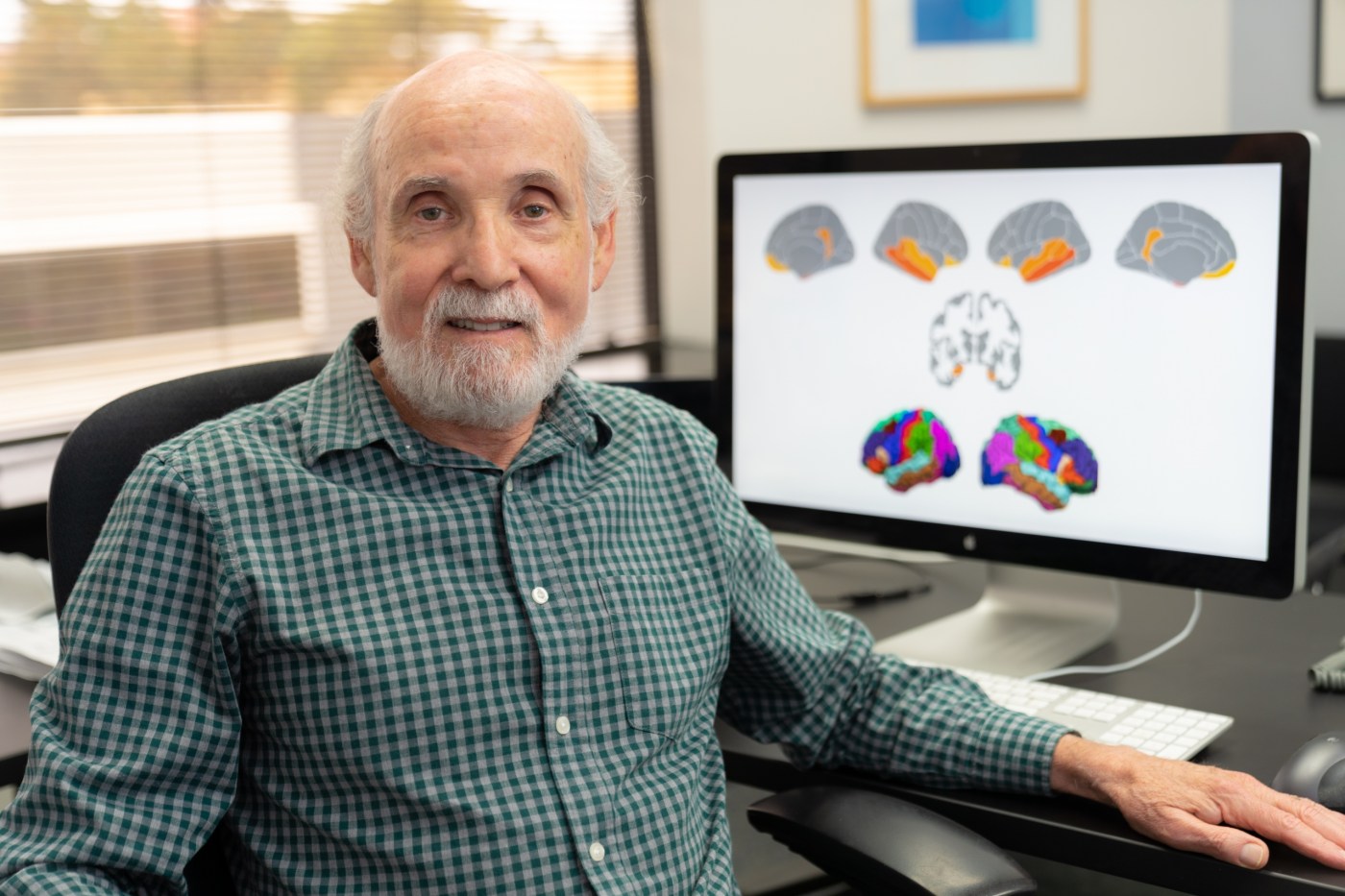
The study included 169 cognitively normal male Veterans with an average age of 56. In the main finding, Kremen and his team learned that a loss of structural integrity, as indicated by the images, significantly improved the prediction of progression to mild cognitive impairment over a 12-year follow-up period. A loss of structural integrity in brain tissue can be a sign of neurodegeneration.
The research team, which used magnetic resonance imaging (MRI) to produce the brain images, based its finding “on a continuous measure to observe the degree of structural integrity from better to worse,” Kremen notes. “We’re not at the stage yet where we can have a set cutoff for abnormality. It’s an index of what’s called microstructural integrity. Our results suggest that poorer structural integrity tends to be associated with a higher risk for developing mild cognitive impairment later on.”
In a secondary analysis, Kremen and his colleagues looked at brain images while controlling for the effects of general aging. They observed that the Alzheimer’s-related brain regions remained as significant predictors, suggesting that the effect was not simply due to general aging.
The researchers used grey matter images to predict the progression to mild cognitive impairment. Grey matter is a key component of the central nervous system that helps people control movement, memory and emotions. Grey matter images, the research team believed, would be better predictors of mild cognitive impairment than those based on cortical thickness or the volume of the hippocampus.
A novel brain signature
Cortical thickness is a measure used to describe the combined thickness of layers of the cerebral cortex, which is involved in complex brain functions, such as language and information processing. The hippocampus is a brain region that is important for its involvement in memory. One of the most consistent features of Alzheimer’s disease is atrophy in certain brain regions, particularly the hippocampus. Prior research, Kremen says, has used images based on measurements of grey matter thickness and volume.
In Kremen’s study, the researchers used an Alzheimer’s brain signature that they previously developed with data from the Alzheimer’s Disease Neuroimaging Initiative, a multisite study that aims to improve clinical trials for preventing and treating Alzheimer’s. The signature, a weighted average of thickness in seven cortical regions plus the volume of the hippocampus, was published about a decade ago in a study led by Dr. Linda McEvoy, a scientist at the University of California, San Diego. She was a co-author of Kremen’s 2021 paper.
Kremen’s team then created a novel brain signature based on grey matter diffusivity. Mean diffusivity is the average mobility of water molecules in the brain tissue.
More Information
Click here to read the full story.
Click here to learn more about VA research.
Topics in this story
More Stories
Diverse representation of women in health care research allows MVP to make discoveries for women’s health
Join the Million Veteran Program online. You will have the option to receive an at-home blood sample collection kit in the mail.
VHA's new podcast series, New Horizons in Health, features a candid discussion of psychedelic assisted therapies for Veterans experiencing mental health conditions.






I would like to see if I can qualify for this study. I live in Orlando Florida
Thanks for your inquiries. It is not possible to enroll in our study because it only includes twins in the already established Vietnam Era Twin Study of Aging. However, you could check to see if there is Alzheimer’s disease research at the VA in your area or look for an Alzheimer’s Disease Research Center in your area. There are many Alzheimer’s Disease Research Centers around the country. Both places and also Alzheimer’s treatment centers or memory clinics are places you can go to for evaluation and care. I also want to be clear that our work described in the article is still in the research phase and is not yet ready to be directly applied for clinical evaluation.
Bill Kremen
Hello,
I have a cognitive issue and disangnosed and want to see how this could help.