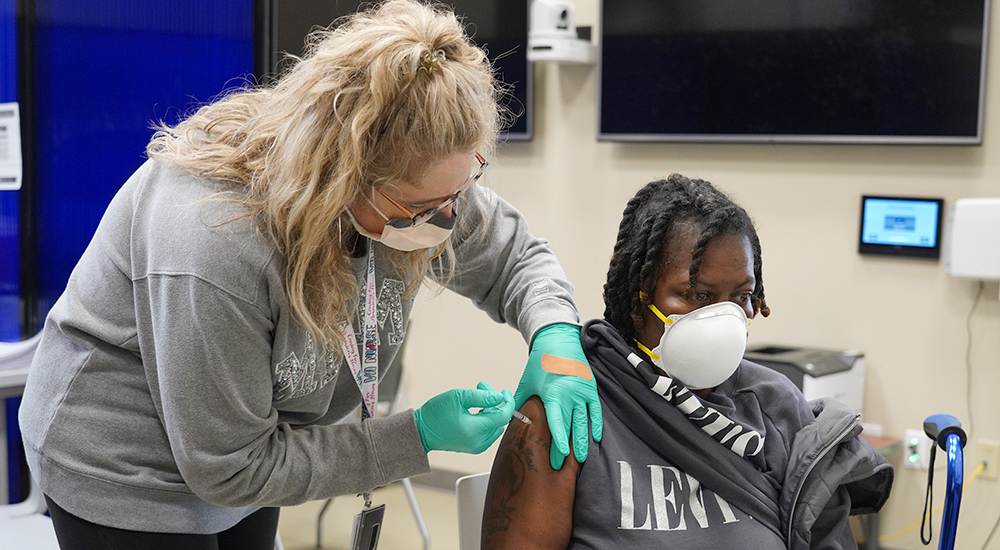
VA received 71,400 doses of the Janssen COVID-19 vaccine, March 3, authorized by the U.S. Food and Drug Administration under an Emergency Use Authorization.
In clinical trials, the Janssen vaccine showed an efficacy of 66% against moderate to severe/critical COVID-19 disease occurring at least 28 days after vaccination – and demonstrated greater than 85% efficacy in preventing severe/critical COVID-19 disease occurring at least 28 days after vaccination.
“VA now has a third highly effective vaccine to support the department’s critical efforts in vaccinating Veterans,” said Acting VA Under Secretary for Health Richard Stone, M.D. “This one-dose vaccine will help us vaccinate more people faster and will help us effectively reach more Veterans, including those in rural areas.”
As of March 3, VA provided Pfizer-BioNTech and Moderna COVID-19 vaccine first doses to more than 1,648,000 Veterans, employees and federal partners – and both doses to more than 925,000 of these individuals.
Veterans who are enrolled and receiving health care in VA are eligible to get the vaccine when their facility has vaccine supply and reaches their risk category. Veterans are required to enroll with VA in order to receive health care. However, to receive care in VA, enrollees must meet certain eligibility requirements under current law, which may include income limits.
Veterans can get the latest information and sign up to receive updates on VA’s COVID-19 vaccine webpage.
Topics in this story
More Stories
Seven U.S. Army soldiers, one Army Reserve soldier and two Veterans are representing Team USA at the 2024 Olympic Games in Paris, which begins today.
Study underscores important role COVID vaccination can have in protecting Veterans from infection and reducing long-term health consequences
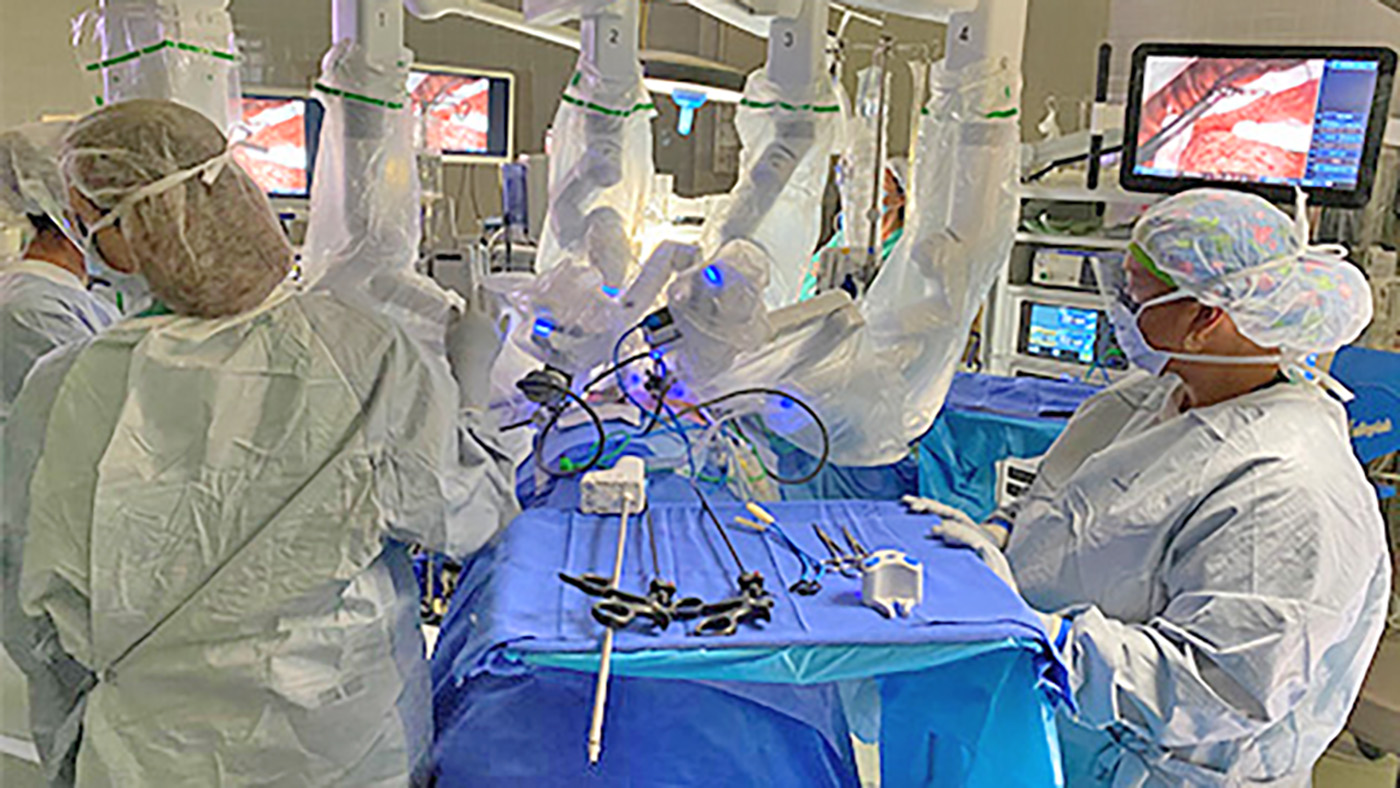
Columbia VA’s robotic surgery teams completed their 800th robotic surgery and are on schedule to hit 1,000 by the end of the year.





va in st petersburg fl. and other nearby va facilities would not give me a covid vaccine because I was only 67. I applied to every county within 50 miles. Just got my second shot of moderna.
I called the VAMC here in Albuquerque and asked for the Jassen version and they said their allocation already went to veterans who were at “high risk”.
When was a vaccine called Janssen approved and why hasn’t this been reported on mainstream news?
Seriously? We are the test subjects. Before they release it or any other 1-time dose alternatives, they have to test it first. It has an 85% efficacy rate which by comparison is much lower than the 2-dose alternative currently being given by Moderna and Pfizer which have efficacy rates of 95% and 94.1%.
If you Google the Jaansen Vaccine, you will find that it is the name used by Johnson & Johnson for its vaccine. The information provided is correct. It is true that people don’t recognize this name because the media doesn’t use it, but that doesn’t make the VA wrong.
It’s the Johnson & Johnson (J&J) vaccine. Janssen is the name of their vaccine research division (which I believe is based in Europe). I’m guessing we called it Johnson & Johnson in the US is because either we wanted to claim credit for it or because Americans are more likely to trust a vaccine from a familiar company.
I am 83 years old. I received the Moderna vaccine at the VA in Madison, Wisconsin. First shot was Jan. 16, second was Feb 18. Both visits could not have gone smoother. These VA people in Madison are ORGANIZED !
Thank you so much VA
Imagine being in a foreign country that has the only VA Clinic outside the US or it’s territories. The VA Clinic says any minute now and the vaccine will be here while at the same time the US Embassy says the vaccine will not be made available to any US citizens outside the US. When the US Embassy is asked to explain the why two different things are being published they do not respond. Please do not respond to this message, just imagine.
Recieved my first dose at VA in North Chicago and was told spouses were not eligible yet. When will that be?
My husband received his first dose of the vaccine at the Capt. James Lovell VA in North Chicago, was told spouses were not eligible yet. When will it be available for spouses?
To Whom It May Concern:
You have the name of the vaccine maker wrong!
It is the Johnson and Johnson (JNJ) vaccine that was recently approved by the FDA.
Please be more careful in reporting vital VA information to veterans and please ensure you state just the correct and accurate facts.
We are all very tired of the constant Trump and Trumpite lies and misinformation and disinformation.
Thanks,
Dan
When will the out patient clinics in Florida get more vaccines? I’m disabled, and told that I have to drive an hour to get it at VA hospital.
Also – when will the one dose Johnson and Johnson vaccine be available to veterans in Florida?
What nonsense. Stop wasting time and resources with this ridiculous propaganda b.s. and try answering your damn phones for a change.
I would like to know when the VAMC Togus in Maine will start receiving the Johnson & Johnson vaccine.
As of yesterday, only 9.97% of Ohioans have received a full Covid vaccination (both shots). I can’t believe how slow it’s going!
I moved to San Antonio, Texas in November 2019 from O’ahu, Hawaii, where my wife and I were both enrolled in the Tripler Army Medical Center system for our PRIMARY medical care. When I arrived in San Antonio, Texas VA was already shut down to new enrollees and I was denied care through JBSA active-duty medical resources.
The VA system took my registration but it was April 2020 before I could even get registered in the Southwest Texas VA system. I was forced to seek civilian care via the Medicare system over telemedicine to obtain access to my THIRTEEN prescriptions reauthorized until I could obtain an appointment to actually SEE a LIVE PROVIDER !! I have recently changed my Medicare plan administrator to Humana HMO and they reassigned me and my wife to see a NEW doctor.
I have had the most difficult time obtaining an appointment to receive ANY COVID vaccine through the VA. I am 72 years old with four major comorbidities (HTN, Type 2 Diabetes, Kidney disease, obesity) but I have Medicare parts A, B, and D and my primary provider is a civilian Internist via my Humana HMO provider. VA provides my Apnea care and supplies but will not give me access to their immunization process because they are not my primary medical source !!
Calls to the VA appointment system are maddening. The main scheduling number is constantly BUSY and hangs up immediately. I have ventured to the clinic I am assigned to but all appointments are booked solid through March. They have suggested other satellite clinics over 20 miles away, but those numbers also go unanswered after navigating their ridiculous screening messages. TEXAS is a very irrational state in its political response to COVID policies and I am fearful of contracting the virus if I’m forced to wait any longer to be vaccinated !!
Where can I turn to for the J&J COVIT vaccine? How can I get scheduled??
Houston VA is a couple of hours away and they are providing COVID-19 immunizations to veterans of all ages as of this week. I hope this helps. You should have been immunized by now.
Pretty Sure there’s a Typo, in that second paragraph
I would like to find out when and how I can make an appointment to get the Johnson & Johnson COVID vaccination. I live in Palm Desert, Ca 92260
When will JNJ vaccine be available at the American Lake VA facility in WA State
hOW DO i REGISTER FOR A COVID 19 VACCINE?
I am a 100 percent P&T veteran located in the Atlanta, GA area. I have re’cd both Pfizer shots from a local health department. My who serves as my caretaker wants the Johnson and Johnson vaccine single shot. Thank you for your assistance.
Can you let me know where I can get the Johnson and johnson vaccination shot in san diego
Good afternoon,
I would like to know when the VA Haborhealth Care Center in NYC will start receiving the Johnson & Johnson vaccine.
Thanks for your help!
Josephine