COVID-19 is an urgent health crisis that has demanded the attention of top scientific experts. To date, researchers have developed a handful of new therapies to treat COVID-19 patients. Unfortunately, the sickest individuals often succumb to respiratory failure, organ failure or death. Worldwide 4.9 million people have died from the disease.
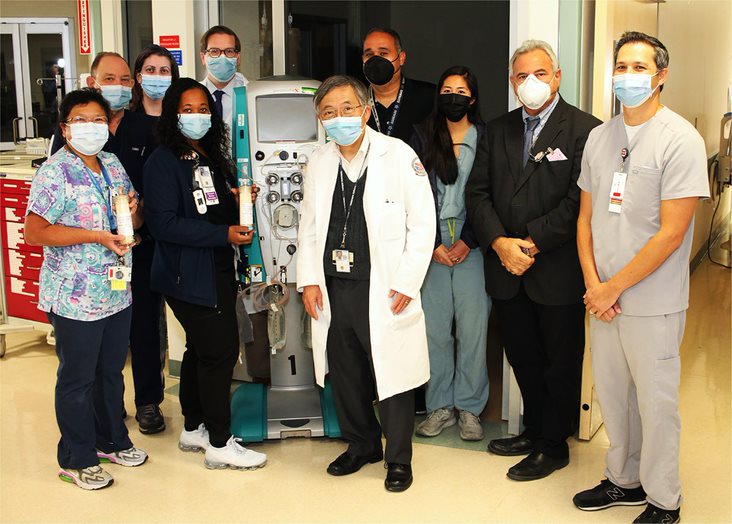
Now, scientists at the Department of Defense, VA and partner institutions are testing a new technology to treat COVID-19. It’s called the Seraph 100 blood filter. The treatment uses a novel approach to filter out viral particles and harmful molecules from the bloodstream of COVID-19 patients.
Dr. Kevin Chung is principal investigator for the PURIFY OBS clinical trial that will examine the effectiveness of the Seraph filter. He is chair of the department of medicine at the Uniformed Services University in Bethesda, Maryland.
“The Seraph filter is different because it targets the pathogen (SARS-CoV-2) to prevent a maladaptive inflammatory response in the body, which itself can cause harm. We think disease severity from COVID-19 correlates with the body’s response to the virus. What we are trying to do with Seraph is to remove that trigger,” says Chung. “I liken it to shutting off the faucet, instead of mopping up the floor.”
Rigorous clinical testing
The PURIFY program is a four-part, multi-site clinical evaluation that involves 13 participating sites across the U.S, including one VA medical center. The first stage of PURIFY is a retrospective, observational study that aims to review the safety and effectiveness of the Seraph 100 filter to treat severe COVID-19. Physicians will follow 90 patients with severe COVID-19 who received usual care and treatment with the Seraph filter. The team will compare outcomes in this group with those of an equal number of COVID-19 patients who were treated with usual care alone.
The research team posted preliminary data for the first stage (PURIFY-OBS), in April 2021. They plan to submit the complete data for publication soon. Physicians initially treated 53 COVID-19 patients with the Seraph filter at four ICUs. Severely ill patients who were treated with the Seraph filter experienced a 38% mortality rate, compared with a 67% mortality rate for patients not treated with the new technology. Only one patient treated with the Seraph filter experienced an adverse event – an episode of low blood pressure that responded to medication.
The second stage of PURIFY is a prospective study and will involve 30 patients. Physicians will take blood samples before and after treatment with the Seraph filter to characterize viral load and inflammation markers.
Stage 3 of PURIFY (which has two parts) includes a feasibility study and a randomized clinical trial to determine the effectiveness of the Seraph filter for treating all pathogen-related shock – not just shock caused by COVID-19.
Sickest COVID-19 patients can experience life-threatening lung failure
Shock is a serious medical condition where cells in the body do not receive enough oxygen to sustain function. It can result in organ failure and death. There are many conditions that can lead to shock. Examples are traumatic injury, hemorrhage and infection. Septic shock is most often caused by bacterial infections that set off an exaggerated immune response in the body.
In the case of COVID-19, the sickest patients can experience severe acute respiratory syndrome (SARS), which involves life-threatening lung failure. If a patient is given that diagnosis in COVID-19, that means their likelihood of dying is very high, says Chung.
The Seraph filter received an emergency use authorization (EUA) from the Food and Drug Administration in April 2020. Treatment with the Seraph filter is currently reserved for intensive-care patients with COVID-19 who are in, or close to, respiratory failure. Researchers hope to get full FDA approval for the Seraph filter so that it can be commercialized and made available for broader applications.
More Information
Click here to read the full story.
Click here to learn more about VA research.
Click here to read about VA research during the COVID-19 pandemic.
Topics in this story
More Stories
Updated COVID vaccines are available free of charge to Veterans receiving care at VA .
It can take up to a couple of weeks for a vaccine to fully protect you, so be sure to get both your flu and COVID-19 vaccines soon.
Receiving an updated COVID-19 vaccine increases the immune response, which improves protection from becoming seriously ill from COVID-19.