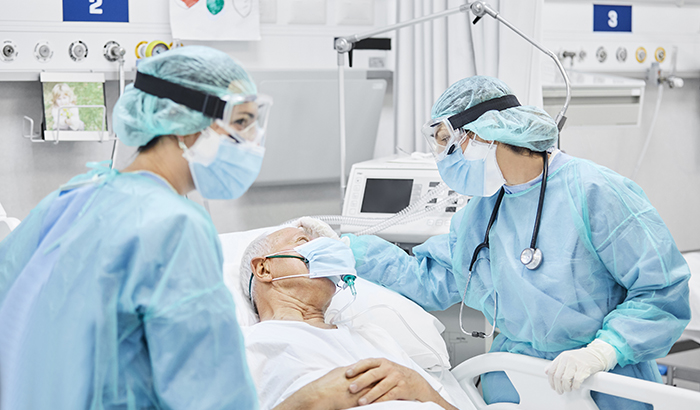
VA is launching a new clinical trial to study convalescent plasma for treating seriously ill COVID-19 patients.
Convalescent plasma is the liquid part of the blood that contains antibodies, which are proteins the body makes to fight infections, such as COVID-19. It is donated by people who have recovered from COVID-19.
“This trial will go a long way toward helping in the fight against COVID-19,” VA Secretary Robert Wilkie said in a press release. “VA CURES will provide valuable information that will benefit our Veterans who are battling COVID-19, as well as other patients and the medical community in general.”
700 Veterans to take part in trial
Some 700 Veterans who are hospitalized at VA medical centers are being enrolled in the trial, with half getting convalescent plasma and the other half in a control group. The key outcome measurement is the number of patients who died from any cause or had respiratory failure by day 28. The main secondary outcome is time to recovery by day 28.
Earlier this year, the U.S. Food and Drug Administration (FDA) authorized use of convalescent plasma as an investigational treatment for COVID-19 through the FDA’s expanded access program. That program was used widely throughout the nation, including about 80 VA sites, as an approach for seriously ill patients who have few, if any, options left. It ended last month. The FDA has now authorized emergency use of the therapy, based on the available scientific evidence to date. That means not only can researchers use convalescent plasma for investigational purposes, but health care providers can also use it for hospitalized patients with COVID-19.

Dr. Edward Janoff, an infectious disease specialist at the VA Eastern Colorado Health Care System, is co-leading the first trial on convalescent plasma. (Photo by Terri Rorke)
VA initiative to give Veterans faster access to COVID-19 trials
The trial is the first of multiple studies in VA CURES, a new initiative that is aimed at giving Veterans faster access to trials for potential COVID-19 treatments and at confirming their effectiveness. VA CURES—the “VA Coronavirus Research and Efficacy Studies”—is a master protocol that offers a standardized framework for studies on many potential treatments for COVID-19, without the need for a new study design and protocol each time. Over the coming months, VA CURES studies will evaluate drugs for different stages of COVID-19 infection, from the prevention to outpatient and inpatient stages.
About 25 VA medical centers are participating in VA CURES. The program is led by infectious disease, critical care, and pulmonary experts in VA.
“This coordinated effort for controlled trials is very important so we can develop an infrastructure within VA to show what works with our patients,” says Dr. Edward Janoff, an infectious disease specialist at the VA Eastern Colorado Health Care System.
`That’s our immediate priority’
VA CURES studies will evaluate new treatments, such as antiviral agents and drugs that impact the immune system. Some trials may include outpatients with mild symptoms to prevent their progression to hospitalization. The VA CURES team also plans to prioritize preventive measures for Veterans who would be at high risk for illness should they become infected.
“We will begin by carefully evaluating treatments to study that we think have the best chance of bringing about an effective treatment not only for Veterans but also for other people,” says Dr. Sheldon Brown, an infectious disease specialist at the James J. Peters VA Medical Center in the Bronx, New York. “That’s our immediate priority. The only thing we can rely on is good quality evidence. That’s exactly the purpose of having a program like VA CURES. We want to obtain the best quality evidence we possibly can so people are not misled about a benefit or the potential harm of a treatment when, in fact, it may be something that’s important to help them.”
More Information
Click here to read the full story.
Click here for more information on VA COVID-19 research.
Click here to learn more about VA research.
Topics in this story
More Stories
Seven U.S. Army soldiers, one Army Reserve soldier and two Veterans are representing Team USA at the 2024 Olympic Games in Paris, which begins today.
The findings of this new MVP study underscore the importance and positive impact of diverse representation in genetic research, paving the way for significant advances in health care tailored to Veteran population-specific needs.
VA reduces complexity for Veterans, beneficiaries, and caregivers signing in to VA.gov, VA’s official mobile app, and other VA online services while continuing to secure Veteran data.